UFC 3-570-06
JANUARY 31 2003
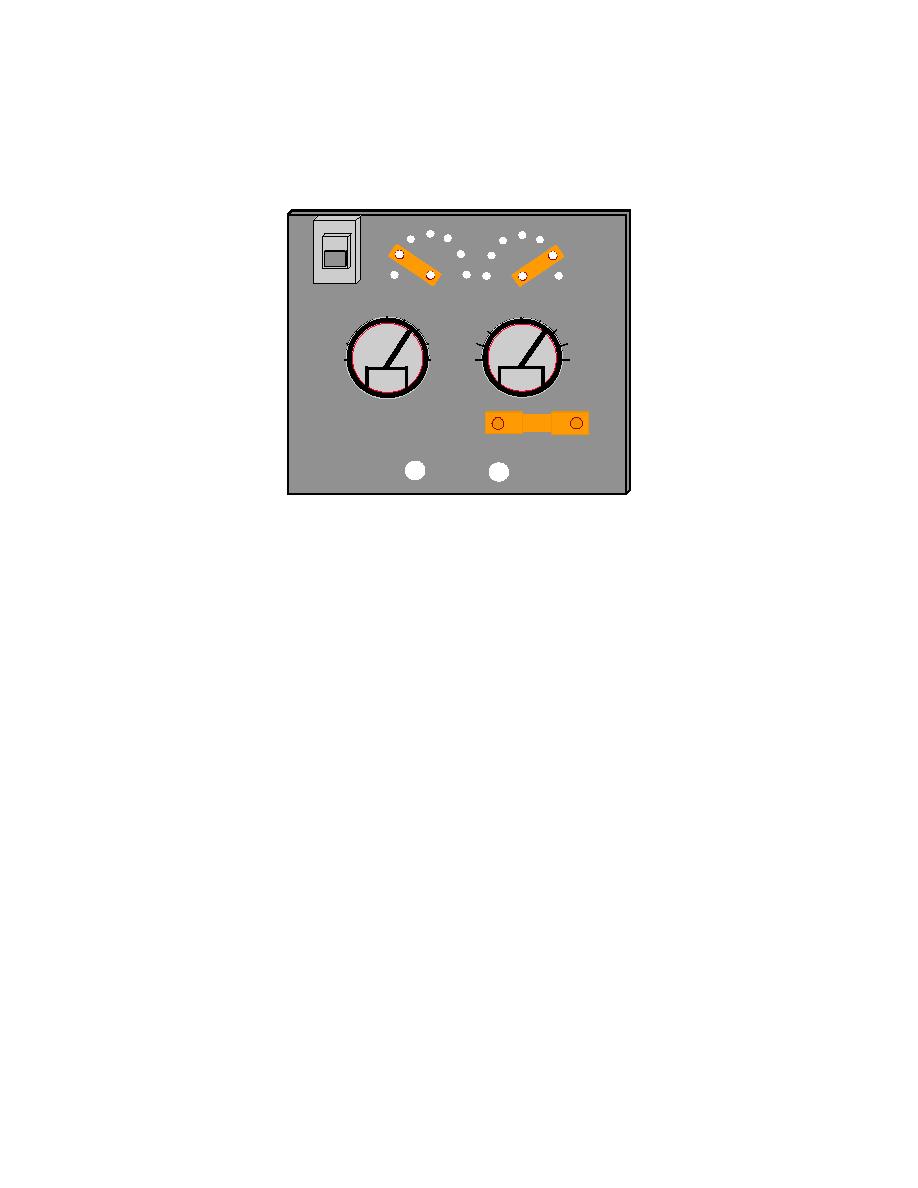
Figure 2-25. Impressed Current Cathodic Protection System Rectifier
4
8
5
10
3
6
6
12
2
4
1
7
2
14
8
0
16
0
-
+
2-7.2
Impressed Current Anode Materials. The anodes of an impressed current
system provide the means for the protective current to enter the electrolyte. Since the
anodes form the corroding part of the system, the best material is one that has a low
rate of weight loss per ampere-year (Table 2-11). The most commonly used materials
for impressed current anodes are graphite and high-silicon cast iron. Aluminum is
sometimes used in water storage tanks. In areas where heavy icing is not a problem,
high silicon cast iron anodes are used instead of aluminum for lower long-term cost.
Platinum coated titanium or niobium anodes and mixed metal oxide anodes are
becoming more prevalent as impressed current anode material.
Before the 1970s, there were only three types of anodes primarily used for
impressed current groundbeds--high silicon cast iron, graphite, and scrap steel. As
technology progressed, so did the types of materials that are commonly used by
corrosion personnel. The materials have specialized applications and, when installation
and operating costs are assessed, very few anodes can be used universally for any
type of application and still achieve a desirable design life. In most soils, anodes evolve
oxygen and the anode oxidizes as the current is discharged. In chloride containing soils
or water, anodes evolve chlorine gas that forms hydrochloric acid, and the anodes
break down chemically. The whole purpose of this explanation is to point out that some
anodes perform well in the presence of oxygen and others in the presence of acids.
2-44




 Previous Page
Previous Page
